Our pipeline
is a strategic bridge
Between cutting-edge
science
and real-world
impact


True to our vision, our robust pipeline serves as a bridge between cutting-edge science and real-world impact—designed to deliver differentiated and globally relevant healthcare advancements.
Through Ichnos Glenmark Innovation (IGI), we are focused on addressing some of the most critical unmet needs to ensure accessibility to patients across the globe.

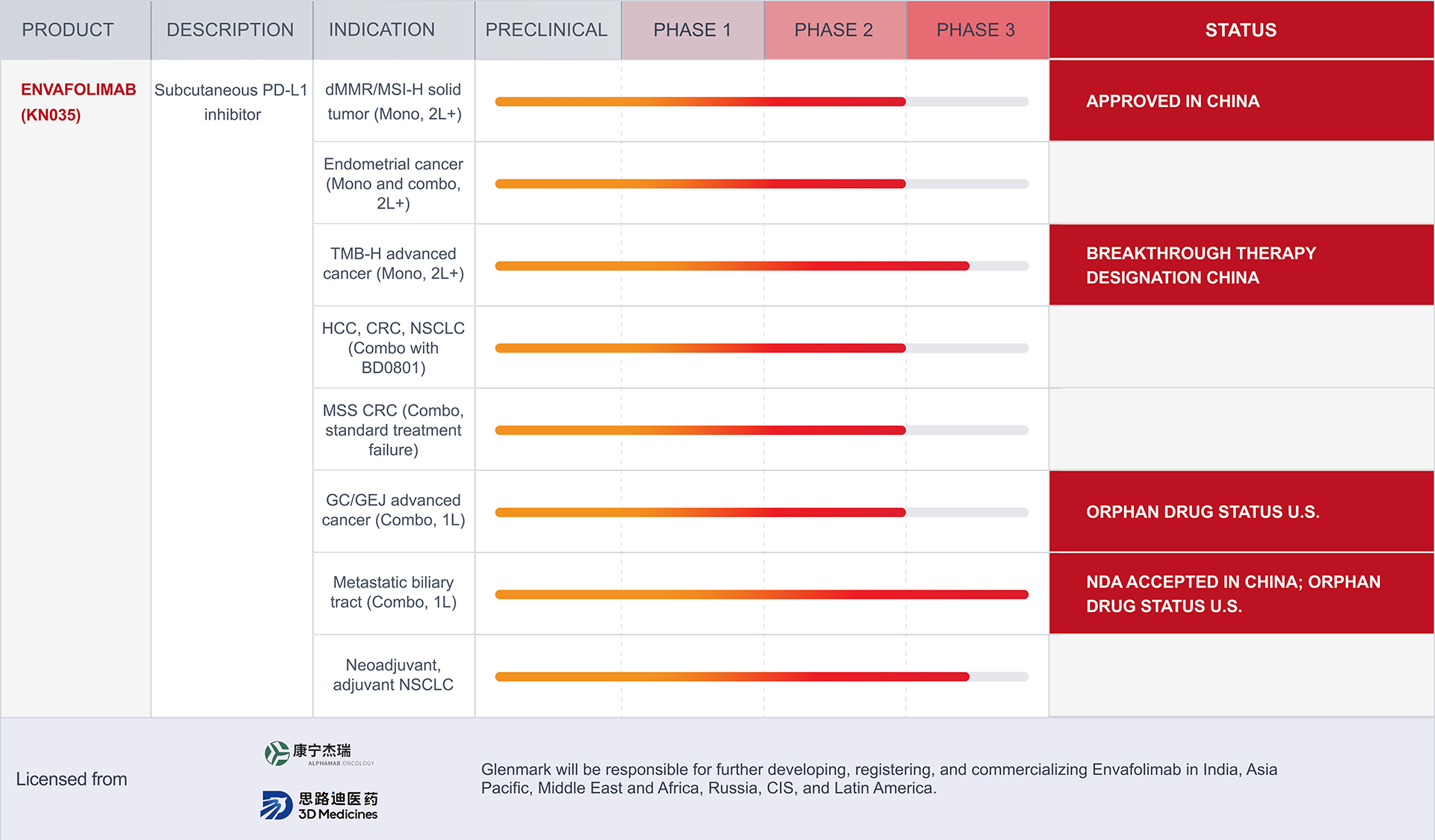
dMMR: Mismatch Repair Deficient
MSI-H: Microsatellite Instability-High
TMB-H: High Tumor Mutational Burden
HCC: Hepatocellular Carcinoma
CRC: Colorectal Cancer
NSCLC: Non-Small Cell Lung Cancer
MSS: Microsatellite Stability
GC/GEJ: Gastric and Gastroesophageal Junction Cancer
Disclaimer:
The pipeline displayed above includes therapeutic candidates for which Glenmark has in-licensed rights to develop and/or commercialize. These programs may be pursued by Glenmark independently or with its partners. The portfolio reflects the status of such in-licensed assets as of April 5, 2026, and is subject to change. As these assets originate externally, their inclusion in the pipeline reflects Glenmark’s contractual development and/or commercialization rights and does not indicate internal discovery by Glenmark. Program progress depends on multiple factors, including pre-clinical and clinical studies, regulatory requirements, scientific evaluation, and market conditions. As a result, the pipeline is not exhaustive, and programs may advance, be delayed, paused, or discontinued over time. Nothing on this page should be interpreted as a guarantee of future approval, availability, or commercialization.
This page provides general information only and is not a substitute for medical advice. Please consult a qualified medical practitioner for diagnosis and treatment based on your individual condition. Although Glenmark makes reasonable efforts to keep this information current, it may become outdated over time. Glenmark makes no representations or warranties as to the accuracy, completeness, or usefulness of this information and assumes no obligation or liability to update it.
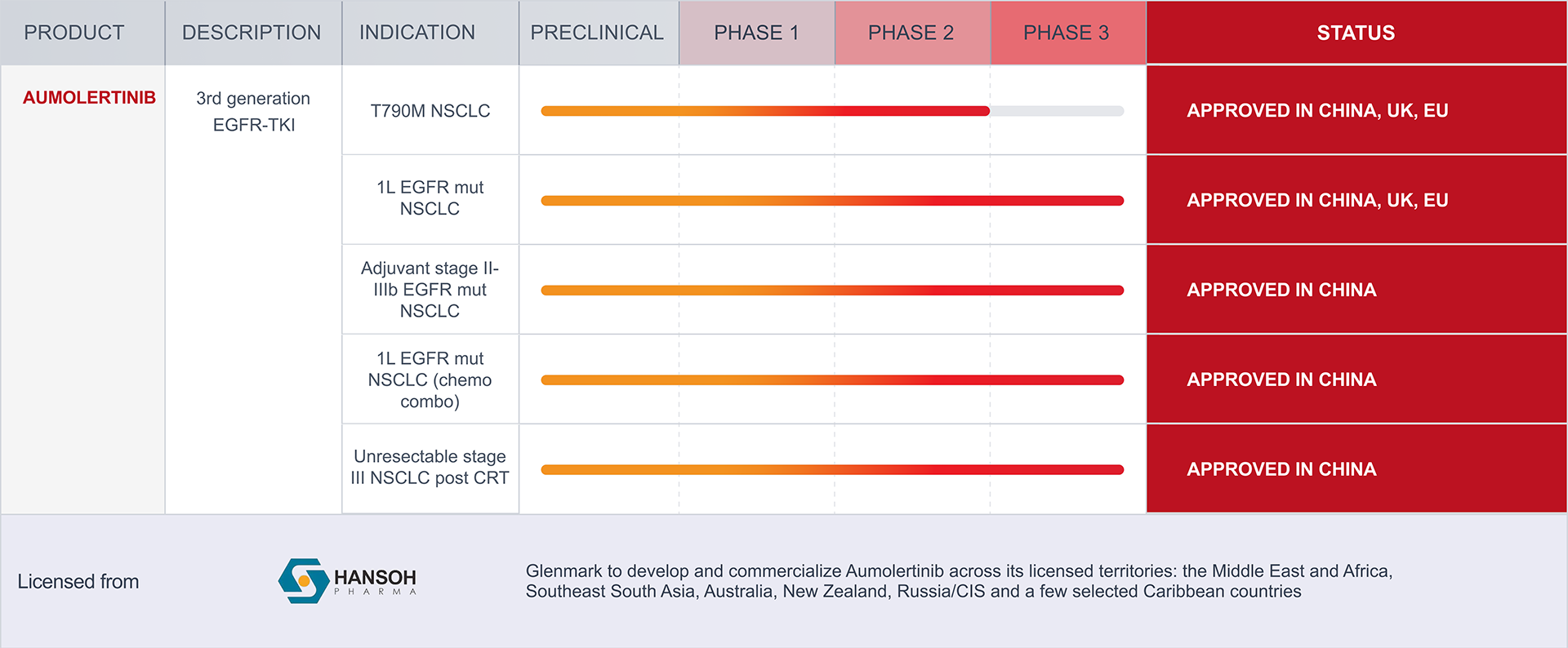
NSCLC: Non-Small Cell Lung Cancer
EGFR: Epidermal Growth Factor Receptor
CRT: Chemoradiotherapy
Disclaimer:
The pipeline displayed above includes therapeutic candidates for which Glenmark has in-licensed rights to develop and/or commercialize. These programs may be pursued by Glenmark independently or with its partners. The portfolio reflects the status of such in-licensed assets as of April 5, 2026, and is subject to change. As these assets originate externally, their inclusion in the pipeline reflects Glenmark’s contractual development and/or commercialization rights and does not indicate internal discovery by Glenmark. Program progress depends on multiple factors, including pre-clinical and clinical studies, regulatory requirements, scientific evaluation, and market conditions. As a result, the pipeline is not exhaustive, and programs may advance, be delayed, paused, or discontinued over time. Nothing on this page should be interpreted as a guarantee of future approval, availability, or commercialization.
This page provides general information only and is not a substitute for medical advice. Please consult a qualified medical practitioner for diagnosis and treatment based on your individual condition. Although Glenmark makes reasonable efforts to keep this information current, it may become outdated over time. Glenmark makes no representations or warranties as to the accuracy, completeness, or usefulness of this information and assumes no obligation or liability to update it.
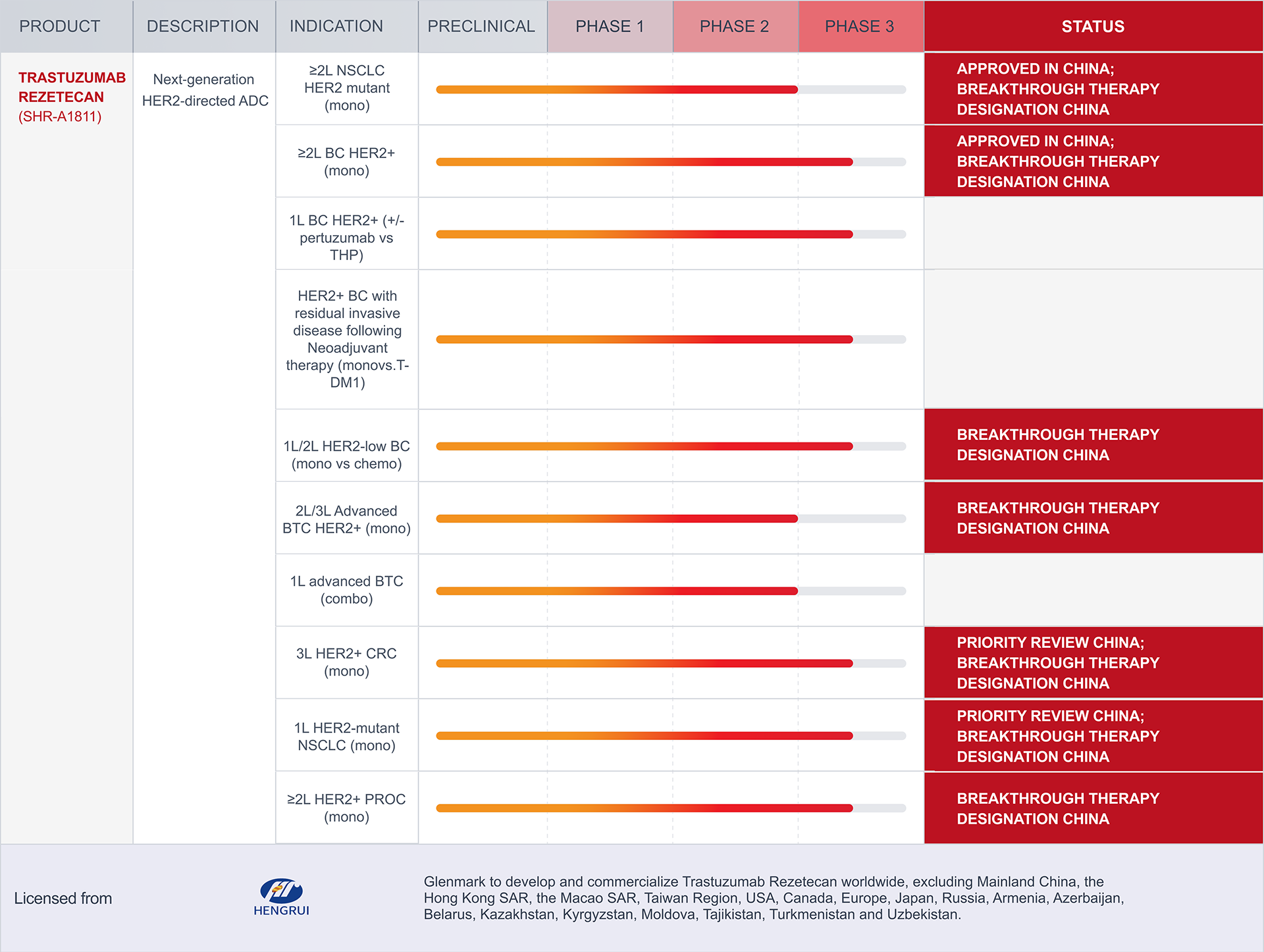
NSCLC: Non-Small Cell Lung Cancer
HER2: Human Epidermal Growth Factor Receptor 2
BC: Breast Cancer
T-DM1: Trastuzumab Emtansine
BTC: Biliary Tract Cancer
GC/GEJC: Gastric and Gastroesophageal Junction Cancer
CRC: Colorectal Cancer
PROC: Platinum-resistant ovarian cancer
THP: Docetael+Trastuzumab+Pertuzumab
Disclaimer:
The pipeline displayed above includes therapeutic candidates for which Glenmark has in-licensed rights to develop and/or commercialize. These programs may be pursued by Glenmark independently or with its partners. The portfolio reflects the status of such in-licensed assets as of April 5, 2026, and is subject to change. As these assets originate externally, their inclusion in the pipeline reflects Glenmark’s contractual development and/or commercialization rights and does not indicate internal discovery by Glenmark. Program progress depends on multiple factors, including pre-clinical and clinical studies, regulatory requirements, scientific evaluation, and market conditions. As a result, the pipeline is not exhaustive, and programs may advance, be delayed, paused, or discontinued over time. Nothing on this page should be interpreted as a guarantee of future approval, availability, or commercialization.
This page provides general information only and is not a substitute for medical advice. Please consult a qualified medical practitioner for diagnosis and treatment based on your individual condition. Although Glenmark makes reasonable efforts to keep this information current, it may become outdated over time. Glenmark makes no representations or warranties as to the accuracy, completeness, or usefulness of this information and assumes no obligation or liability to update it.

