Cutting-edge
R&D capabilities
Pioneering differentiated
innovation-focused therapies
We have built strong capabilities spanning early discovery to late-phase development led by the leading scientists across our six global R&D centres. Spread across India, Europe, and the U.S., this globally integrated network advances complex generics, respiratory innovation, biosimilars, and next generation biologics.
Our research ecosystem is designed to scale impactful science, accelerate differentiated therapies, and enable faster, more efficient global launches.
We conduct clinical trials that are scientifically rigorous, ethically sound and globally compliant. Every clinical study we undertake is built on the pillars of patient safety, scientific integrity, and regulatory excellence.
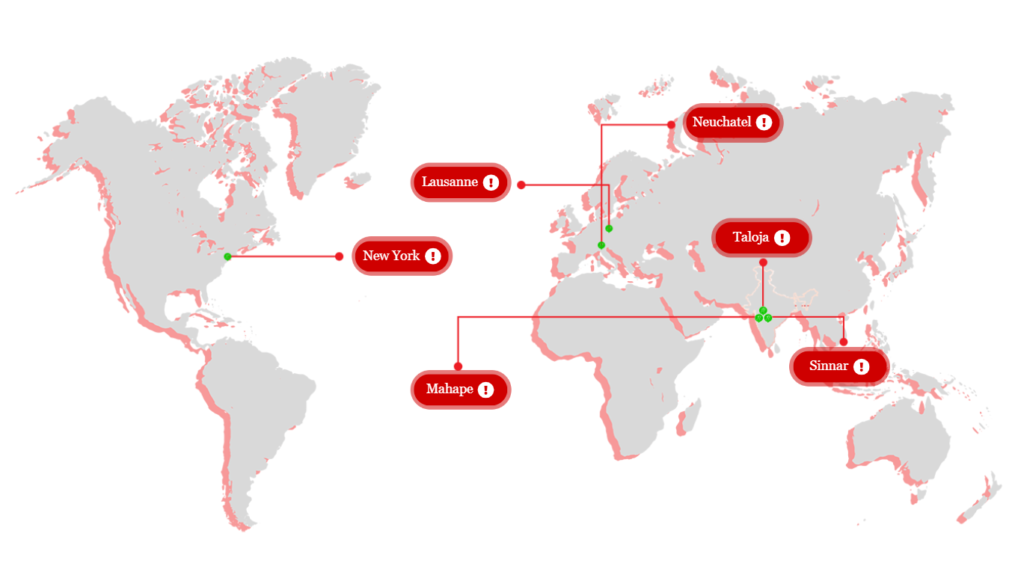
Our global R&D facilities

Sinnar
We established our first R&D center in Sinnar, Maharashtra, India.
It focuses on formulations development and analytical research of specialty and branded formulations for the global markets. The research is based on dosage forms for therapeutic areas like respiratory, dermatology, oncology, cardiovascular, anti- diabetes, etc., and facilitates research of aerosols of various indications (metered dose inhalers, dry powder inhalers, nebulizers and nasal sprays). It is equipped with state-of-the-art equipment and technologies, ensuring innovative and effective formulation solutions. The facility is also ISO 14001:2015 certified.
Capabilities:
Equipped with state-of-the-art equipment and technologies, ensuring innovative and effective formulation solutions. ISO 14001:2015 certified.
Taloja
Our R&D centre in Taloja, Mumbai, India is Focus on developing cost-effective and patent-driven generic formulations. The centre specializes in Diverse Dosage Forms: Oral solids, semi-solids, Parenteral (complex injectables), Drug Device and Aerosols.
It also houses an advanced clinical research & pharmacokinetics facility with 60 Clinical Beds, ICU, Bioanalytical Lab for
BA/BE studies.
Capabilities:
• Advanced Analytical Division: HPLC, LCMS, and NGI for formulation development.
• Leading Aerosol Research: MDIs and DPIs, top
experimental apparatus.
• Advanced Clinical Research & Pharmacokinetics facility: 60 Clinical Beds, ICU, Bioanalytical Lab for BA/BE studies.
• ISO 14001:2015 certified
Lausanne
As the heart of our biologics innovation, our Research Center in Lausanne focuses on the discovery and development of biologics and is home IGI’s groundbreaking BEAT® platform. It has End-to-end capabilities in discovering and developing Multispecifics™ from inception through preclinical and Specialization clinical studies.
Capabilities:
Our team conducts the scientific investigation supporting the clinical development of our product candidates, translating basic research into potential new therapies that may one day benefit patients.
Neuchatel
The centre leads the strategic development and value creation for our Specialty and Innovative Business portfolio. The team oversees global registrations, clinical development, quality and commercialization support for small molecules, biologics and ADCs in the pipeline.
Capabilities
Advanced competencies across multiple global functions with deep expertise in navigating complex regulatory and clinical development landscape across developed and emerging markets, in order to drive accessibility of next-generation innovative therapies to patients in developed markets and high-burden underserved geographies.
Mahape
Our Mahape facility also functions as the small molecule research centre for IGI. Together IGI Antibody Discovery, Pharmacokinetics and Toxicology teams support selection and progression of safe and effective biologic drugs into to clinical trials that may one day benefit patients providing targeted and novel treatment options.
Capabilities:
• Leveraging sophisticated instruments like Franz diffusion cell, HPLC, LCMS etc.
• Together IGI Antibody Discovery, Pharmacokinetics and Toxicology teams support selection and progression of safe and effective biologic drugs into to clinical trials that may one day benefit patients providing targeted and novel treatment options.
New York
The centre in New York leads strategic direction of IGI and drives the clinical development of its multispecific™ and small-molecule pipeline. The team oversees IND-enabling activities and early clinical development through value inflection and out-licensing.
Capabilities:
Deep expertise in advancing innovative therapies through complex regulatory and competitive landscapes, translating science into treatments for patients with high unmet medical need.
*Map is for representational purpose only
*Map is for representational purpose only
Lausanne
As the heart of our biologics innovation, our Research Center in Lausanne focuses on the discovery and development of biologics and is home IGI’s groundbreaking BEAT® platform. It has End-to-end capabilities in discovering and developing Multispecifics™ from inception through preclinical and Specialization clinical studies.
Capabilities:
Our team conducts the scientific investigation supporting the clinical development of our product candidates, translating basic research into potential new therapies that may one day benefit patients.
New York
The centre in New York leads strategic direction of IGI and drives the clinical development of its multispecific™ and small-molecule pipeline. The team oversees IND-enabling activities and early clinical development through value inflection and out-licensing.
Capabilities:
Deep expertise in advancing innovative therapies through complex regulatory and competitive landscapes, translating science into treatments for patients with high unmet medical need.
Mahape
Our Mahape facility also functions as the small molecule research centre for IGI. Together IGI Antibody Discovery, Pharmacokinetics and Toxicology teams support selection and progression of safe and effective biologic drugs into to clinical trials that may one day benefit patients providing targeted and novel treatment options.
Capabilities:
• Leveraging sophisticated instruments like Franz diffusion cell, HPLC, LCMS etc.
• Together IGI Antibody Discovery, Pharmacokinetics and Toxicology teams support selection and progression of safe and effective biologic drugs into to clinical trials that may one day benefit patients providing targeted and novel treatment options.
Neuchatel
The centre leads the strategic development and value creation for our Specialty and Innovative Business portfolio. The team oversees global registrations, clinical development, quality and commercialization support for small molecules, biologics and ADCs in the pipeline.
Capabilities
Advanced competencies across multiple global functions with deep expertise in navigating complex regulatory and clinical development landscape across developed and emerging markets, in order to drive accessibility of next-generation innovative therapies to patients in developed markets and high-burden underserved geographies.
Taloja
Our R&D centre in Taloja, Mumbai, India is Focus on developing cost-effective and patent-driven generic formulations. The centre specializes in Diverse Dosage Forms: Oral solids, semi-solids, Parenteral (complex injectables), Drug Device and Aerosols.
It also houses an advanced clinical research & pharmacokinetics facility with 60 Clinical Beds, ICU, Bioanalytical Lab for
BA/BE studies.
Capabilities:
• Advanced Analytical Division: HPLC, LCMS, and NGI for formulation development.
• Leading Aerosol Research: MDIs and DPIs, top
experimental apparatus.
• Advanced Clinical Research & Pharmacokinetics facility: 60 Clinical Beds, ICU, Bioanalytical Lab for BA/BE studies.
• ISO 14001:2015 certified
Sinnar
We established our first R&D center in Sinnar, Maharashtra, India.
It focuses on formulations development and analytical research of specialty and branded formulations for the global markets. The research is based on dosage forms for therapeutic areas like respiratory, dermatology, oncology, cardiovascular, anti- diabetes, etc., and facilitates research of aerosols of various indications (metered dose inhalers, dry powder inhalers, nebulizers and nasal sprays). It is equipped with state-of-the-art equipment and technologies, ensuring innovative and effective formulation solutions. The facility is also ISO 14001:2015 certified.
Capabilities:
Equipped with state-of-the-art equipment and technologies, ensuring innovative and effective formulation solutions. ISO 14001:2015 certified.

